
For example, the XeF 2 molecule has a steric number of five and a trigonal bipyramidal geometry. With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. For some molecules in the Table, we note that there is more than one possible shape that would satisfy the VSEPR rules. The F-Br-F bond angle is 86.2, and the Br-F bond lengths are 172 pm and 181 pm respectively.  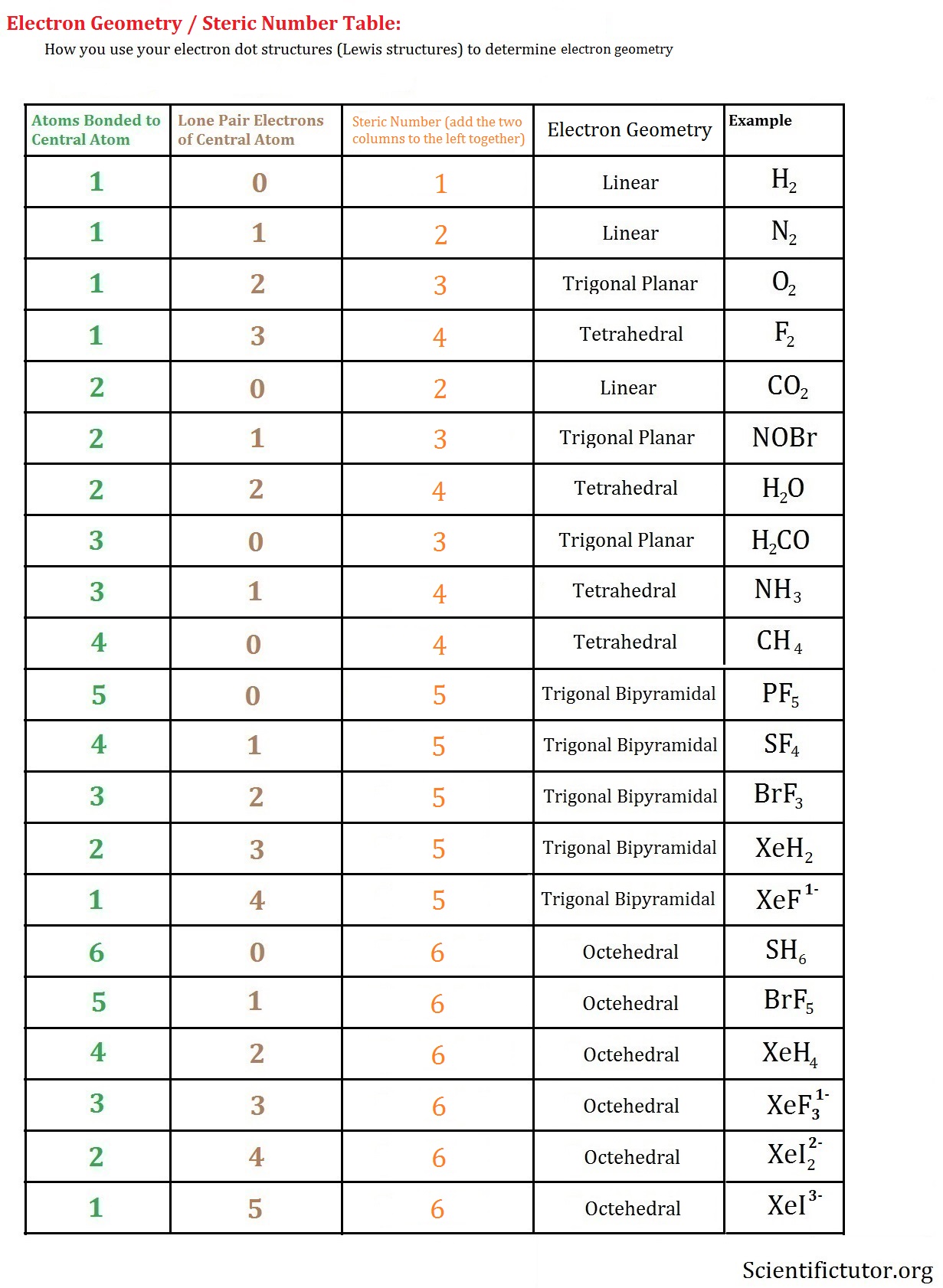
The central Br atom is sp 3 d hybridized in BrF 3. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. It is due to the presence of 2 lone pairs on the central bromine atom in the BrF 3 molecule that it adopts a distorted shape, different from its ideal electron pair geometry. egetrigonal planar, mgtrigonal planar o egtrigonal bipyramidal. Start your trial now First week only 4.99 arrow. Having a straw i.e, colorless to yellow appearance, this chemical compound has several applications but also comes with a number of limitations and hazard issues. Brf3 molecular geometry name Electron Pair Geometry - BYJU N2 Molecular Geometry - BRAINGITH WebThe best place to start when trying to figure out a. electron affinity., If a central atom is surrounded by six electron domains, the term used to describe its fundamental geometry is a. BrF3, known as Bromine Trifluoride, is a fuming liquid consisting of inter-halogen combinations and bearing a pungent smell. Shape of Molecules Electron Pair geometry helps. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. Question: Determine the electron geometry (eg) and molecular geometry (mg) of BrF3. Solution for Predict the electron-domain and molecular geometries of(a) BrF3, (b) SF5+. Study with Quizlet and memorize flashcards containing terms like The overall shape of a molecule is determined by its a. Brf3 Lewis Structure Molecular Geometry The total valence electron is 28 for drawing BrF3 Lewis structure. We designate SF 4 as AX 4E it has a total of five electron pairs. In PF 5, axial atoms (2 P-F bonds) make a 90º angle with the plane, and equatorial atoms (3 P-F bonds) make a 120º angle with each other. The Lewis structure of a compound gives specific insight into its molecular geometry and shape. Each fluorine atoms has nine electrons, and there are seven valence electrons in the outer shell of the Bromine molecule, out of which three electrons form bonds with three fluorine atoms. Study with Quizlet and memorize flashcards containing terms like Determine the electron geometry (eg) and molecular geometry (mg) of BrF3., Determine the electron geometry (eg), molecular geometry (mg), and polarity of SF6, Give the approximate bond angle for a molecule with tetrahedral electron geometry and bent molecular geometry. Upon adding the remaining two Chlorine atoms, we end up with a Trigonal Bipyramidal geometry with bond angles of 90° & 120°. BrF3 is a perfect example of an AX5 molecule with two lone pairs of electrons and three bonded pairs of electrons. 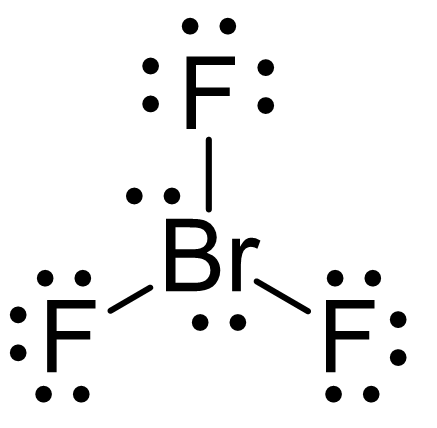
The hybridization of phosphorous in PF 5 is sp 3 d. At three Chlorine atoms, we get Trigonal Planar molecular geometry. The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.\).ģ. The electron geometry of PF 5 is also Trigonal bipyramidal. VSEPR Theory: a chemistry model used to predict the shape of individual molecules based on electron-pair electrostatic repulsion.The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral.Molecular geometries take into account the number of atoms and the number of lone pair electrons.Fundamentally, the VSEPR model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Molecular Geometry Notation for BrF3 Molecule : Is BCl3 trigonal planar The molecular geometry of BCl3 is trigonal planar.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
